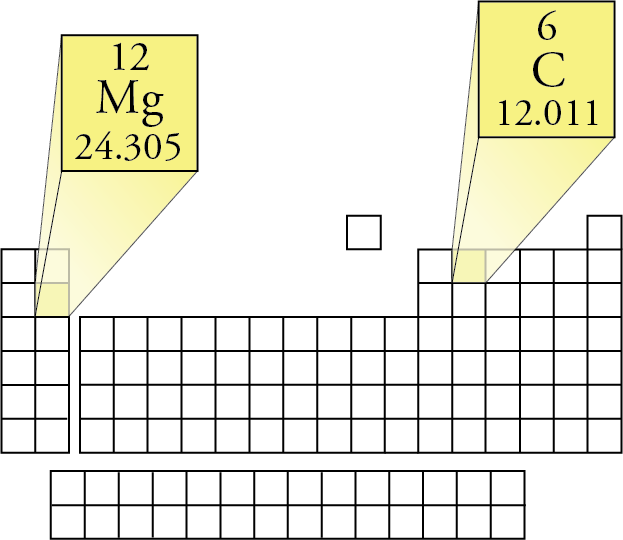
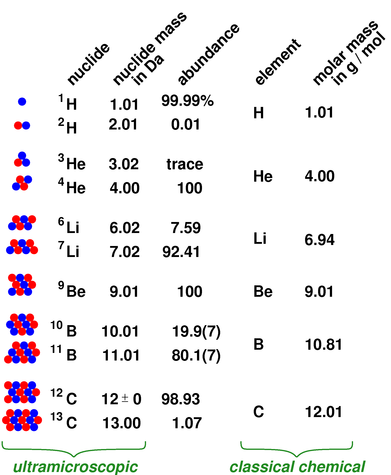
The molecular weight of water is 18 grams. Molecular weight of one water molecule is calculated, when we add (2 x 1) + 16 = 18 grams. In addition, the atomic weight of hydrogen is 1 gram and oxygen is 16 grams. Water has 3 atoms, two hydrogen atoms, and one oxygen atom. 18 grams per mole is the molar mass of water. We calculate the molar mass of one water molecule, when we add (2 x 1) + 16 = 18 grams. Also, the atomic weight of hydrogen is 1 gram and oxygen is 16 grams. It is calculated as the mass of substance relative to the 1/12 th of the mass of the carbon-12 atom. It is calculated by dividing the mass of a substance by the amount of the substance 1: Moles to Mass Conversion with Elements. According to the periodic table, 1 mol of U has a mass of 238.0 g, so the mass of 2 mol is twice that, or 476.0 g. Some elements are usually encountered as molecules, e.g. It is the mass of a mole of a particular substance The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. Multiplying by the molar mass constant ensures that the calculation is dimensionally correct: standard relative atomic masses are dimensionless quantities (i.e., pure numbers) whereas molar masses have units (in this case, grams per mole). Difference Between Molar Mass and Molecular Weight Basis When considering the element, aluminum, we can use the periodic table of elements to look up the atomic weight of this element (26.98 amu). With the help of the above example, we can say that both molar mass and molecular mass are the same in their values but are different in their units. In such a conversion, we use the molar mass of a substance as a conversion factor to convert mole units into mass units (or, conversely, mass units into mole units). Therefore the molecular weight of H 2O = (1x 99 amu) + (1 x 35.45 amu).The number of chlorine atoms present = 1.In other words, we can say that the Molecular weight of a particular molecule is equal to the sum of the atomic masses of each element. With the help of the above equation, we can find the molecular weight of an element or a compound. Add them together: add the results from step 3 to get the total molar mass of the compound. Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound. Therefore, the unit of the molecular weight taken as atomic mass units or amu. The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). The molecular weight has no units because the division has done between two masses that have the same units. Molar mass is the mass of a mole of a particular substance means the mass of a particular compound divided by the amount of substance. While the definition and units are different for molar mass and molecular weight, the value is the same. Whereas molecular weight is the mass of a molecule of a particular substance. Moreover, the main difference between both is that molar mass gives the mass of a mole of a particular substance. Besides, the molar mass and molecular weight are two physical properties of substances.Īlmost all chemical reactions are involved the changes occur in molecules or compounds so it is very important to understand the chemical and physical properties of molecules in order to perform chemical reactions and assume the final products. These molecules attached to each other to form compounds. Molecules are substances formed due to combinations of the same or different atoms in different ratios. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals.Difference Between Molar Mass & Molecular Weight Please note that the elements do not show their natural relation towards each other as in the Periodic system.

The unity for atomic mass is gram per mol.

The lightest chemical element is Hydrogen and the heaviest is Hassium. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight). This list contains the 118 elements of chemistry.
Plant Inspection & Process OptimalisationĬhemical elements listed by atomic mass The elemenents of the periodic table sorted by atomic massĬlick on any element's name for further information on chemical properties, environmental data or health effects.

